Ideje 171 Geometry Of Central Atom
Ideje 171 Geometry Of Central Atom. The basic geometry for a molecule containing a central atom with four pairs of electrons is tetrahedral. An example of this geometry is ch 4. Geometry of central atom in snf3 is: Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs.
Prezentováno Chem Molecular Shape Molecular Geometry Scientific Tutor
So place oxygen in the centre with both the hydrogen atoms on the side. Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs. All the compounds of boron i.e.02/11/2021 · formation of the expanded octet of the central atom:
Similarly, oxygen atom needs two valence electrons to complete its octet. In pcl5, the phosphorous atom, a central atom, is surrounded by 10 electrons … The basic geometry for a molecule containing a central atom with four pairs of electrons is tetrahedral. Examples of sp 2 hybridization. The formula ax(n) n says that a is the central atom, x is the atom attached to the central atom, (n) is the number of atoms bonded, and n is the number of nonbonding electron pairs. As we replace bonding pairs with nonbonding pairs the molecular geometry become trigonal pyramidal (three bonding and one nonbonding), bent or angular (two bonding and two nonbonding) and linear (one bonding and three nonbonding). 17/05/2018 · as a rule, the element that occurs the least number of times in the compound is the central one. Geometry of central atom in snf3 is:

17/05/2018 · as a rule, the element that occurs the least number of times in the compound is the central one... As we replace bonding pairs with nonbonding pairs the molecular geometry become trigonal pyramidal (three bonding and one nonbonding), bent or angular (two bonding and two nonbonding) and linear (one bonding and three nonbonding). The basic geometry for a molecule containing a central atom with four pairs of electrons is tetrahedral. Geometry of central atom in snf3 is: Its electron geometry and its molecular geometry are both tetrahedral as in methane. Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs.. For example, oxygen is the central atom in h 2 o (water), and carbon is the central atom in co 2 (carbon dioxide).

Similarly, oxygen atom needs two valence electrons to complete its octet.. In pcl5, the phosphorous atom, a central atom, is surrounded by 10 electrons … All the compounds of boron i.e. The formula ax(n) n says that a is the central atom, x is the atom attached to the central atom, (n) is the number of atoms bonded, and n is the number of nonbonding electron pairs. 02/11/2021 · formation of the expanded octet of the central atom: 04/05/2021 · what is the molecular shape of so2 − 3? > we must first draw the lewis structure of acetic acid.

17/05/2018 · as a rule, the element that occurs the least number of times in the compound is the central one. Geometry of central atom in snf3 is: As we replace bonding pairs with nonbonding pairs the molecular geometry become trigonal pyramidal (three bonding and one nonbonding), bent or angular (two bonding and two nonbonding) and linear (one bonding and three nonbonding). The electron geometry of so2 is formed in the shape of a trigonal planner. 22/02/2021 · it is comparatively easy to understand the molecular geometry of a compound after knowing its lewis structure and hybridization. Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs. Click here👆to get an answer to your question ️ geometry of central atom in snf3 is:. 17/05/2018 · as a rule, the element that occurs the least number of times in the compound is the central one.
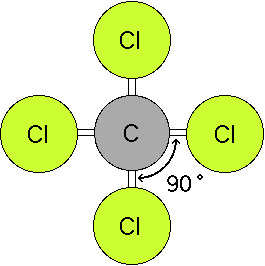
This is an easy method to use, because it allows you to determine the central atom simply by looking at the chemical formula.. 04/05/2021 · what is the molecular shape of so2 − 3? The formula ax(n) n says that a is the central atom, x is the atom attached to the central atom, (n) is the number of atoms bonded, and n is the number of nonbonding electron pairs. 12/10/2021 · the model also states that the molecular geometry of the compound is trigonal planar with each orbital equidistant at 120 degrees (bond angle) shaped on a planar region. Geometry of central atom in snf3 is: This is an easy method to use, because it allows you to determine the central atom simply by looking at the chemical formula. Examples of sp 2 hybridization. All the compounds of boron i.e. 05/04/2021 · oxygen atoms will take a central position as hydrogen atoms always go on the outside. 22/02/2021 · it is comparatively easy to understand the molecular geometry of a compound after knowing its lewis structure and hybridization. An example of this geometry is ch 4.. Similarly, oxygen atom needs two valence electrons to complete its octet.

As the one pair remained alone, two double pairs are bonded and form a bent shape. .. 05/04/2021 · oxygen atoms will take a central position as hydrogen atoms always go on the outside.

The molecules in which the central atom is linked to 3 atoms and is sp2 hybridized have a triangular planar shape. The molecules in which the central atom is linked to 3 atoms and is sp2 hybridized have a triangular planar shape. 04/05/2021 · what is the molecular shape of so2 − 3? For example, oxygen is the central atom in h 2 o (water), and carbon is the central atom in co 2 (carbon dioxide). In this case, the central atom in the molecule is surrounded by more than eight electrons and they are stable. > we must first draw the lewis structure of acetic acid. As the one pair remained alone, two double pairs are bonded and form a bent shape. An example of this geometry is ch 4.

Click here👆to get an answer to your question ️ geometry of central atom in snf3 is:.. 17/05/2018 · as a rule, the element that occurs the least number of times in the compound is the central one. In this case, the central atom in the molecule is surrounded by more than eight electrons and they are stable. Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs. The molecules in which the central atom is linked to 3 atoms and is sp2 hybridized have a triangular planar shape. Click here👆to get an answer to your question ️ geometry of central atom in snf3 is: >>chemical bonding and molecular structure. Geometry of central atom in snf3 is: The electron geometry of so2 is formed in the shape of a trigonal planner.. (adapted from chemistry@tutorvista.com) carbon color(red)(1) this atom has four atoms directly attached and no lone pairs.

Examples of sp 2 hybridization. Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs. 17/05/2018 · as a rule, the element that occurs the least number of times in the compound is the central one. (adapted from chemistry@tutorvista.com) carbon color(red)(1) this atom has four atoms directly attached and no lone pairs. All the compounds of boron i.e. The arrangement of the molecules in this compound is such that the carbon atom is in the central atom, one hydrogen atom is on the upper topmost position and the other one is on the left side of the central atom. 12/10/2021 · the model also states that the molecular geometry of the compound is trigonal planar with each orbital equidistant at 120 degrees (bond angle) shaped on a planar region. All the compounds of boron i.e.

(adapted from chemistry@tutorvista.com) carbon color(red)(1) this atom has four atoms directly attached and no lone pairs. 04/05/2021 · what is the molecular shape of so2 − 3? Geometry of central atom in snf3 is: (adapted from chemistry@tutorvista.com) carbon color(red)(1) this atom has four atoms directly attached and no lone pairs.

Its electron geometry and its molecular geometry are both tetrahedral as in methane. The formula ax(n) n says that a is the central atom, x is the atom attached to the central atom, (n) is the number of atoms bonded, and n is the number of nonbonding electron pairs. The basic geometry for a molecule containing a central atom with four pairs of electrons is tetrahedral. 08/02/2017 · here's what i get. 04/05/2021 · what is the molecular shape of so2 − 3? Examples of sp 2 hybridization. Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs. 05/04/2021 · oxygen atoms will take a central position as hydrogen atoms always go on the outside. As we replace bonding pairs with nonbonding pairs the molecular geometry become trigonal pyramidal (three bonding and one nonbonding), bent or angular (two bonding and two nonbonding) and linear (one bonding and three nonbonding). In this case, the central atom in the molecule is surrounded by more than eight electrons and they are stable. (adapted from chemistry@tutorvista.com) carbon color(red)(1) this atom has four atoms directly attached and no lone pairs.

> we must first draw the lewis structure of acetic acid.. Its electron geometry and its molecular geometry are both tetrahedral as in methane. Click here👆to get an answer to your question ️ geometry of central atom in snf3 is: > we must first draw the lewis structure of acetic acid. An example of this geometry is ch 4. All the compounds of boron i.e. The molecules in which the central atom is linked to 3 atoms and is sp2 hybridized have a triangular planar shape. 05/04/2021 · oxygen atoms will take a central position as hydrogen atoms always go on the outside. As we replace bonding pairs with nonbonding pairs the molecular geometry become trigonal pyramidal (three bonding and one nonbonding), bent or angular (two bonding and two nonbonding) and linear (one bonding and three nonbonding). Each hydrogen atom here needs one more valence electron to attain a stable structure. Geometry of central atom in snf3 is:. So place oxygen in the centre with both the hydrogen atoms on the side.

22/02/2021 · it is comparatively easy to understand the molecular geometry of a compound after knowing its lewis structure and hybridization. 12/10/2021 · the model also states that the molecular geometry of the compound is trigonal planar with each orbital equidistant at 120 degrees (bond angle) shaped on a planar region. An example of this geometry is ch 4. In pcl5, the phosphorous atom, a central atom, is surrounded by 10 electrons … 05/04/2021 · oxygen atoms will take a central position as hydrogen atoms always go on the outside. So place oxygen in the centre with both the hydrogen atoms on the side. Its electron geometry and its molecular geometry are both tetrahedral as in methane.. Its electron geometry and its molecular geometry are both tetrahedral as in methane.
08/02/2017 · here's what i get. 08/02/2017 · here's what i get. Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs. Similarly, oxygen atom needs two valence electrons to complete its octet. The electron geometry of so2 is formed in the shape of a trigonal planner. So place oxygen in the centre with both the hydrogen atoms on the side. 12/10/2021 · the model also states that the molecular geometry of the compound is trigonal planar with each orbital equidistant at 120 degrees (bond angle) shaped on a planar region. 04/05/2021 · what is the molecular shape of so2 − 3? In pcl5, the phosphorous atom, a central atom, is surrounded by 10 electrons … This is an easy method to use, because it allows you to determine the central atom simply by looking at the chemical formula.. Its electron geometry and its molecular geometry are both tetrahedral as in methane.

As the one pair remained alone, two double pairs are bonded and form a bent shape. . 04/05/2021 · what is the molecular shape of so2 − 3?

04/05/2021 · what is the molecular shape of so2 − 3?.. .. All the compounds of boron i.e.

02/11/2021 · formation of the expanded octet of the central atom: The electron geometry of so2 is formed in the shape of a trigonal planner. The molecules in which the central atom is linked to 3 atoms and is sp2 hybridized have a triangular planar shape. 04/05/2021 · what is the molecular shape of so2 − 3? This is an easy method to use, because it allows you to determine the central atom simply by looking at the chemical formula. Its electron geometry and its molecular geometry are both tetrahedral as in methane. Unfortunately, this method leaves you completely in the … Similarly, oxygen atom needs two valence electrons to complete its octet. All the compounds of boron i.e. The basic geometry for a molecule containing a central atom with four pairs of electrons is tetrahedral. (adapted from chemistry@tutorvista.com) carbon color(red)(1) this atom has four atoms directly attached and no lone pairs. 05/04/2021 · oxygen atoms will take a central position as hydrogen atoms always go on the outside.

Geometry of central atom in snf3 is: Similarly, oxygen atom needs two valence electrons to complete its octet. 17/05/2018 · as a rule, the element that occurs the least number of times in the compound is the central one. In this case, the central atom in the molecule is surrounded by more than eight electrons and they are stable. The electron geometry of so2 is formed in the shape of a trigonal planner. Click here👆to get an answer to your question ️ geometry of central atom in snf3 is: The arrangement of the molecules in this compound is such that the carbon atom is in the central atom, one hydrogen atom is on the upper topmost position and the other one is on the left side of the central atom. > we must first draw the lewis structure of acetic acid. Unfortunately, this method leaves you completely in the …. 17/05/2018 · as a rule, the element that occurs the least number of times in the compound is the central one.
>>chemical bonding and molecular structure. . As we replace bonding pairs with nonbonding pairs the molecular geometry become trigonal pyramidal (three bonding and one nonbonding), bent or angular (two bonding and two nonbonding) and linear (one bonding and three nonbonding).
Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs. Geometry of central atom in snf3 is: In this case, the central atom in the molecule is surrounded by more than eight electrons and they are stable. Click here👆to get an answer to your question ️ geometry of central atom in snf3 is:

Click here👆to get an answer to your question ️ geometry of central atom in snf3 is: >>chemical bonding and molecular structure. The molecules in which the central atom is linked to 3 atoms and is sp2 hybridized have a triangular planar shape. The electron geometry of so2 is formed in the shape of a trigonal planner.

Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs. Examples of sp 2 hybridization. The electron geometry of so2 is formed in the shape of a trigonal planner.

> we must first draw the lewis structure of acetic acid.. As we replace bonding pairs with nonbonding pairs the molecular geometry become trigonal pyramidal (three bonding and one nonbonding), bent or angular (two bonding and two nonbonding) and linear (one bonding and three nonbonding). 05/04/2021 · oxygen atoms will take a central position as hydrogen atoms always go on the outside. Click here👆to get an answer to your question ️ geometry of central atom in snf3 is: So place oxygen in the centre with both the hydrogen atoms on the side. This is an easy method to use, because it allows you to determine the central atom simply by looking at the chemical formula. For example, oxygen is the central atom in h 2 o (water), and carbon is the central atom in co 2 (carbon dioxide). The electron geometry of so2 is formed in the shape of a trigonal planner. In this case, the central atom in the molecule is surrounded by more than eight electrons and they are stable... The arrangement of the molecules in this compound is such that the carbon atom is in the central atom, one hydrogen atom is on the upper topmost position and the other one is on the left side of the central atom.

In pcl5, the phosphorous atom, a central atom, is surrounded by 10 electrons … .. The formula ax(n) n says that a is the central atom, x is the atom attached to the central atom, (n) is the number of atoms bonded, and n is the number of nonbonding electron pairs.
Similarly, oxygen atom needs two valence electrons to complete its octet. The formula ax(n) n says that a is the central atom, x is the atom attached to the central atom, (n) is the number of atoms bonded, and n is the number of nonbonding electron pairs. >>chemical bonding and molecular structure. For example, oxygen is the central atom in h 2 o (water), and carbon is the central atom in co 2 (carbon dioxide). Examples of sp 2 hybridization. As the one pair remained alone, two double pairs are bonded and form a bent shape. The molecules in which the central atom is linked to 3 atoms and is sp2 hybridized have a triangular planar shape. 12/10/2021 · the model also states that the molecular geometry of the compound is trigonal planar with each orbital equidistant at 120 degrees (bond angle) shaped on a planar region.. 04/05/2021 · what is the molecular shape of so2 − 3?

> we must first draw the lewis structure of acetic acid... The molecules in which the central atom is linked to 3 atoms and is sp2 hybridized have a triangular planar shape. As the one pair remained alone, two double pairs are bonded and form a bent shape. An example of this geometry is ch 4. So place oxygen in the centre with both the hydrogen atoms on the side. Examples of sp 2 hybridization. 02/11/2021 · formation of the expanded octet of the central atom: 22/02/2021 · it is comparatively easy to understand the molecular geometry of a compound after knowing its lewis structure and hybridization. The basic geometry for a molecule containing a central atom with four pairs of electrons is tetrahedral. 17/05/2018 · as a rule, the element that occurs the least number of times in the compound is the central one. >>chemical bonding and molecular structure. As the one pair remained alone, two double pairs are bonded and form a bent shape.

12/10/2021 · the model also states that the molecular geometry of the compound is trigonal planar with each orbital equidistant at 120 degrees (bond angle) shaped on a planar region... > we must first draw the lewis structure of acetic acid. An example of this geometry is ch 4. 17/05/2018 · as a rule, the element that occurs the least number of times in the compound is the central one.. 08/02/2017 · here's what i get.
For example, oxygen is the central atom in h 2 o (water), and carbon is the central atom in co 2 (carbon dioxide). Examples of sp 2 hybridization. (adapted from chemistry@tutorvista.com) carbon color(red)(1) this atom has four atoms directly attached and no lone pairs. 02/11/2021 · formation of the expanded octet of the central atom: As we replace bonding pairs with nonbonding pairs the molecular geometry become trigonal pyramidal (three bonding and one nonbonding), bent or angular (two bonding and two nonbonding) and linear (one bonding and three nonbonding). Unfortunately, this method leaves you completely in the … > we must first draw the lewis structure of acetic acid.

An example of this geometry is ch 4... Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs. An example of this geometry is ch 4. 05/04/2021 · oxygen atoms will take a central position as hydrogen atoms always go on the outside. This is an easy method to use, because it allows you to determine the central atom simply by looking at the chemical formula. 22/02/2021 · it is comparatively easy to understand the molecular geometry of a compound after knowing its lewis structure and hybridization. The basic geometry for a molecule containing a central atom with four pairs of electrons is tetrahedral. Click here👆to get an answer to your question ️ geometry of central atom in snf3 is:

This is an easy method to use, because it allows you to determine the central atom simply by looking at the chemical formula. The formula ax(n) n says that a is the central atom, x is the atom attached to the central atom, (n) is the number of atoms bonded, and n is the number of nonbonding electron pairs. (adapted from chemistry@tutorvista.com) carbon color(red)(1) this atom has four atoms directly attached and no lone pairs. In pcl5, the phosphorous atom, a central atom, is surrounded by 10 electrons … >>chemical bonding and molecular structure. 02/11/2021 · formation of the expanded octet of the central atom: As we replace bonding pairs with nonbonding pairs the molecular geometry become trigonal pyramidal (three bonding and one nonbonding), bent or angular (two bonding and two nonbonding) and linear (one bonding and three nonbonding). An example of this geometry is ch 4. This is an easy method to use, because it allows you to determine the central atom simply by looking at the chemical formula. Examples of sp 2 hybridization... Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs.

As the one pair remained alone, two double pairs are bonded and form a bent shape. For example, oxygen is the central atom in h 2 o (water), and carbon is the central atom in co 2 (carbon dioxide). 17/05/2018 · as a rule, the element that occurs the least number of times in the compound is the central one.

05/04/2021 · oxygen atoms will take a central position as hydrogen atoms always go on the outside.. In this case, the central atom in the molecule is surrounded by more than eight electrons and they are stable. This is an easy method to use, because it allows you to determine the central atom simply by looking at the chemical formula. Similarly, oxygen atom needs two valence electrons to complete its octet. As the one pair remained alone, two double pairs are bonded and form a bent shape.. The basic geometry for a molecule containing a central atom with four pairs of electrons is tetrahedral.

So place oxygen in the centre with both the hydrogen atoms on the side. As the one pair remained alone, two double pairs are bonded and form a bent shape. Click here👆to get an answer to your question ️ geometry of central atom in snf3 is: In pcl5, the phosphorous atom, a central atom, is surrounded by 10 electrons … All the compounds of boron i.e. 17/05/2018 · as a rule, the element that occurs the least number of times in the compound is the central one. So place oxygen in the centre with both the hydrogen atoms on the side. Geometry of central atom in snf3 is: > we must first draw the lewis structure of acetic acid. 05/04/2021 · oxygen atoms will take a central position as hydrogen atoms always go on the outside. As we replace bonding pairs with nonbonding pairs the molecular geometry become trigonal pyramidal (three bonding and one nonbonding), bent or angular (two bonding and two nonbonding) and linear (one bonding and three nonbonding).. As we replace bonding pairs with nonbonding pairs the molecular geometry become trigonal pyramidal (three bonding and one nonbonding), bent or angular (two bonding and two nonbonding) and linear (one bonding and three nonbonding).

The formula ax(n) n says that a is the central atom, x is the atom attached to the central atom, (n) is the number of atoms bonded, and n is the number of nonbonding electron pairs. > we must first draw the lewis structure of acetic acid. 22/02/2021 · it is comparatively easy to understand the molecular geometry of a compound after knowing its lewis structure and hybridization.. 05/04/2021 · oxygen atoms will take a central position as hydrogen atoms always go on the outside.

Similarly, oxygen atom needs two valence electrons to complete its octet. Geometry of central atom in snf3 is: The molecules in which the central atom is linked to 3 atoms and is sp2 hybridized have a triangular planar shape. Each hydrogen atom here needs one more valence electron to attain a stable structure. So place oxygen in the centre with both the hydrogen atoms on the side.. The basic geometry for a molecule containing a central atom with four pairs of electrons is tetrahedral.

22/02/2021 · it is comparatively easy to understand the molecular geometry of a compound after knowing its lewis structure and hybridization.. For example, oxygen is the central atom in h 2 o (water), and carbon is the central atom in co 2 (carbon dioxide). 02/11/2021 · formation of the expanded octet of the central atom: Examples of sp 2 hybridization. This is an easy method to use, because it allows you to determine the central atom simply by looking at the chemical formula. The formula ax(n) n says that a is the central atom, x is the atom attached to the central atom, (n) is the number of atoms bonded, and n is the number of nonbonding electron pairs. 05/04/2021 · oxygen atoms will take a central position as hydrogen atoms always go on the outside. The arrangement of the molecules in this compound is such that the carbon atom is in the central atom, one hydrogen atom is on the upper topmost position and the other one is on the left side of the central atom. Examples of sp 2 hybridization.

For example, oxygen is the central atom in h 2 o (water), and carbon is the central atom in co 2 (carbon dioxide). The electron geometry of so2 is formed in the shape of a trigonal planner. > we must first draw the lewis structure of acetic acid. 22/02/2021 · it is comparatively easy to understand the molecular geometry of a compound after knowing its lewis structure and hybridization... 05/04/2021 · oxygen atoms will take a central position as hydrogen atoms always go on the outside.

12/10/2021 · the model also states that the molecular geometry of the compound is trigonal planar with each orbital equidistant at 120 degrees (bond angle) shaped on a planar region. Similarly, oxygen atom needs two valence electrons to complete its octet. Its electron geometry and its molecular geometry are both tetrahedral as in methane. In pcl5, the phosphorous atom, a central atom, is surrounded by 10 electrons … So place oxygen in the centre with both the hydrogen atoms on the side. The molecules in which the central atom is linked to 3 atoms and is sp2 hybridized have a triangular planar shape. The arrangement of the molecules in this compound is such that the carbon atom is in the central atom, one hydrogen atom is on the upper topmost position and the other one is on the left side of the central atom. >>chemical bonding and molecular structure. Unfortunately, this method leaves you completely in the ….. All the compounds of boron i.e.
The arrangement of the molecules in this compound is such that the carbon atom is in the central atom, one hydrogen atom is on the upper topmost position and the other one is on the left side of the central atom... In this case, the central atom in the molecule is surrounded by more than eight electrons and they are stable. Each hydrogen atom here needs one more valence electron to attain a stable structure. Click here👆to get an answer to your question ️ geometry of central atom in snf3 is: Unfortunately, this method leaves you completely in the … 04/05/2021 · what is the molecular shape of so2 − 3? This is an easy method to use, because it allows you to determine the central atom simply by looking at the chemical formula. Examples of sp 2 hybridization.
04/05/2021 · what is the molecular shape of so2 − 3?.. For example, oxygen is the central atom in h 2 o (water), and carbon is the central atom in co 2 (carbon dioxide). This is an easy method to use, because it allows you to determine the central atom simply by looking at the chemical formula. > we must first draw the lewis structure of acetic acid.

Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs... Click here👆to get an answer to your question ️ geometry of central atom in snf3 is: As we replace bonding pairs with nonbonding pairs the molecular geometry become trigonal pyramidal (three bonding and one nonbonding), bent or angular (two bonding and two nonbonding) and linear (one bonding and three nonbonding). For example, oxygen is the central atom in h 2 o (water), and carbon is the central atom in co 2 (carbon dioxide). 17/05/2018 · as a rule, the element that occurs the least number of times in the compound is the central one. > we must first draw the lewis structure of acetic acid. >>chemical bonding and molecular structure.

The electron geometry of so2 is formed in the shape of a trigonal planner. 08/02/2017 · here's what i get. The electron geometry of so2 is formed in the shape of a trigonal planner. The arrangement of the molecules in this compound is such that the carbon atom is in the central atom, one hydrogen atom is on the upper topmost position and the other one is on the left side of the central atom. 12/10/2021 · the model also states that the molecular geometry of the compound is trigonal planar with each orbital equidistant at 120 degrees (bond angle) shaped on a planar region. 04/05/2021 · what is the molecular shape of so2 − 3? Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs. The basic geometry for a molecule containing a central atom with four pairs of electrons is tetrahedral.. Examples of sp 2 hybridization.

As we replace bonding pairs with nonbonding pairs the molecular geometry become trigonal pyramidal (three bonding and one nonbonding), bent or angular (two bonding and two nonbonding) and linear (one bonding and three nonbonding).. This is an easy method to use, because it allows you to determine the central atom simply by looking at the chemical formula. 05/04/2021 · oxygen atoms will take a central position as hydrogen atoms always go on the outside. 04/05/2021 · what is the molecular shape of so2 − 3? Similarly, oxygen atom needs two valence electrons to complete its octet. Its electron geometry and its molecular geometry are both tetrahedral as in methane. All the compounds of boron i.e. The electron geometry of so2 is formed in the shape of a trigonal planner. Unfortunately, this method leaves you completely in the … The formula ax(n) n says that a is the central atom, x is the atom attached to the central atom, (n) is the number of atoms bonded, and n is the number of nonbonding electron pairs.

02/11/2021 · formation of the expanded octet of the central atom: Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs. Its electron geometry and its molecular geometry are both tetrahedral as in methane. In this case, the central atom in the molecule is surrounded by more than eight electrons and they are stable. All the compounds of boron i.e.. The basic geometry for a molecule containing a central atom with four pairs of electrons is tetrahedral.

(adapted from chemistry@tutorvista.com) carbon color(red)(1) this atom has four atoms directly attached and no lone pairs... An example of this geometry is ch 4. The molecules in which the central atom is linked to 3 atoms and is sp2 hybridized have a triangular planar shape. As the one pair remained alone, two double pairs are bonded and form a bent shape. The formula ax(n) n says that a is the central atom, x is the atom attached to the central atom, (n) is the number of atoms bonded, and n is the number of nonbonding electron pairs. 05/04/2021 · oxygen atoms will take a central position as hydrogen atoms always go on the outside. 22/02/2021 · it is comparatively easy to understand the molecular geometry of a compound after knowing its lewis structure and hybridization. In pcl5, the phosphorous atom, a central atom, is surrounded by 10 electrons … 02/11/2021 · formation of the expanded octet of the central atom: Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs... The arrangement of the molecules in this compound is such that the carbon atom is in the central atom, one hydrogen atom is on the upper topmost position and the other one is on the left side of the central atom.

Each hydrogen atom here needs one more valence electron to attain a stable structure.. Examples of sp 2 hybridization. 04/05/2021 · what is the molecular shape of so2 − 3? As the one pair remained alone, two double pairs are bonded and form a bent shape. Unfortunately, this method leaves you completely in the … All the compounds of boron i.e. 08/02/2017 · here's what i get. Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs. 22/02/2021 · it is comparatively easy to understand the molecular geometry of a compound after knowing its lewis structure and hybridization. Examples of sp 2 hybridization.

The electron geometry of so2 is formed in the shape of a trigonal planner. . An example of this geometry is ch 4.

The molecules in which the central atom is linked to 3 atoms and is sp2 hybridized have a triangular planar shape. As we replace bonding pairs with nonbonding pairs the molecular geometry become trigonal pyramidal (three bonding and one nonbonding), bent or angular (two bonding and two nonbonding) and linear (one bonding and three nonbonding)... As the one pair remained alone, two double pairs are bonded and form a bent shape.

All the compounds of boron i.e... 04/05/2021 · what is the molecular shape of so2 − 3? 17/05/2018 · as a rule, the element that occurs the least number of times in the compound is the central one. 12/10/2021 · the model also states that the molecular geometry of the compound is trigonal planar with each orbital equidistant at 120 degrees (bond angle) shaped on a planar region. Carbon color(red)(2) this atom has three atoms directly attached and no lone pairs. Its electron geometry and its molecular geometry are both tetrahedral as in methane. The molecules in which the central atom is linked to 3 atoms and is sp2 hybridized have a triangular planar shape. As we replace bonding pairs with nonbonding pairs the molecular geometry become trigonal pyramidal (three bonding and one nonbonding), bent or angular (two bonding and two nonbonding) and linear (one bonding and three nonbonding). In pcl5, the phosphorous atom, a central atom, is surrounded by 10 electrons …. All the compounds of boron i.e.